
Executive summary
ZeusOSA is a non-invasive treatment designed for people with obstructive sleep apnoea (OSA). The device works by gently stimulating muscles in the airway during sleep to help keep it open, without the need for surgery.
The technology has been developed through a clear, structured pathway with support from the NIHR HealthTech Research Centre (HRC) in Cardiovascular and Respiratory Medicine. This has enabled it to advance from early-stage testing to use within NHS settings, and now into a major NIHR-funded clinical trial, the TESLA MICRON UK Trial.
Building on evidence already generated by the HRC-CRM, this trial will evaluate the clinical effectiveness, adherence, and cost-effectiveness of ZeusOSA across NHS sleep centres. The aim is to generate robust evidence to support NHS adoption of this non-invasive, scalable treatment for an underserved patient population.
Clear purpose and why
OSA is a common condition with symptoms that can include breathing stopping and starting, loud snoring and repeated waking throughout the night. The most common treatment for OSA is using a CPAP device - a machine that gently pumps air into a mask you wear over your mouth or nose while you sleep.
Many people find it difficult to use CPAP, and as a result, a large number of patients stop treatment altogether, leaving their condition unmanaged and increasing the risk of serious health problems.
ZeusOSA was developed to offer a simpler, more comfortable alternative that people are more likely to use. The aim is to help more patients stick with treatment, improve their sleep and overall health, and provide a solution that could be used widely across the NHS. This is especially important as demand for sleep services continues to grow and there is a need for treatments that work well in everyday life, not just in clinical settings.
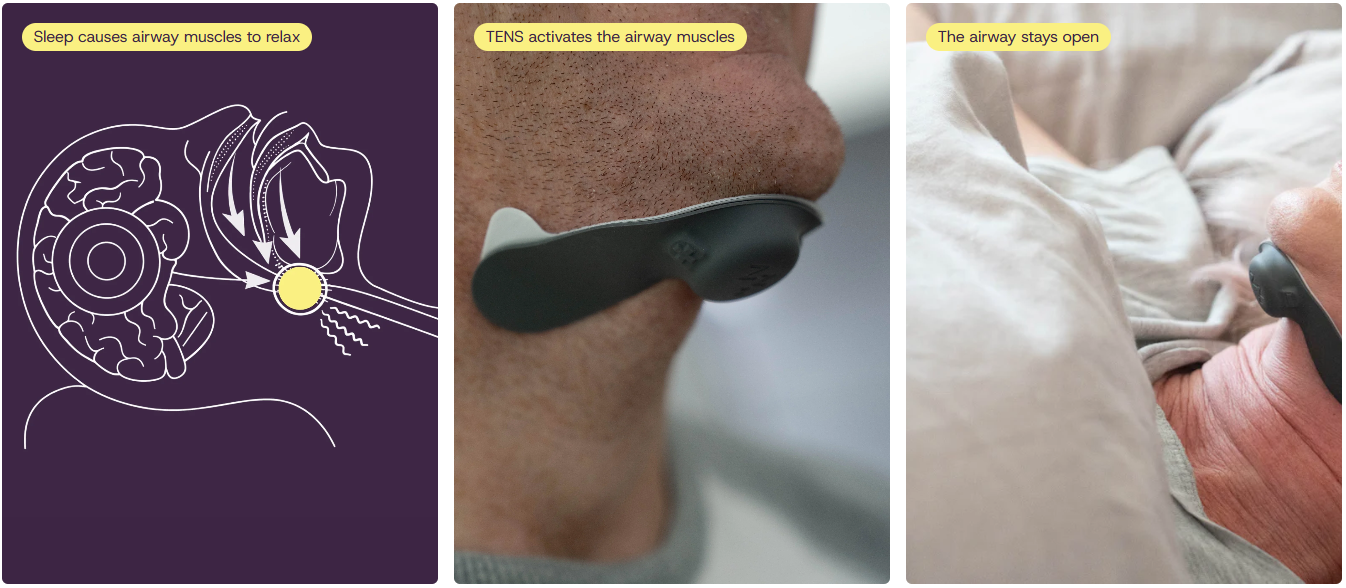
How the device works. Credit: Zeus Sleep Ltd.
Approach
The NIHR HRC-CRM adopts a translational and partnership-driven approach to accelerate the development and evaluation of health technologies within the NHS. By integrating academic research expertise, NHS clinical insight, patient involvement, and industry collaboration, the HRC supports innovations across the full pathway from early feasibility testing to large-scale clinical trials and implementation, including commercialisation and widescale adoption into the NHS.
This approach has been central to the development journey of ZeusOSA. Early studies supported by academic and clinical partners demonstrated the feasibility of transcutaneous hypoglossal nerve stimulation. Subsequent domiciliary and NHS clinical trials generated insights into usability, adherence, and patient outcomes, enabling iterative device optimisation.
Through this structured evidence pathway, the HRC-CRM has supported the progression of ZeusOSA toward definitive evaluation through the TESLA MICRON UK Trial (part of the NIHR Invention for Innovation (i4i) Programme), which aims to generate definitive evidence for clinical effectiveness and NHS implementation.
Insights and impact
The development of ZeusOSA has generated both clinical and real-world insights into the management of OSA. Early clinical studies demonstrated that non-invasive hypoglossal nerve stimulation can be delivered safely and tolerated by patients who struggle with CPAP therapy. Subsequent trials provided important insights into adherence patterns, usability, and patient-reported outcomes, helping refine the technology and inform trial design.
Commercial and consumer adoption has also demonstrated strong demand for non-invasive sleep solutions. Since launch, ZeusOSA has achieved retail distribution with major partners, international sales across 54 countries, and significant media reach, indicating global relevance of the problem addressed.
Reflections
The ZeusOSA development journey demonstrates the importance of combining clinical evidence generation with early market validation. Rapid commercial adoption and strong international demand highlight the global need for alternative therapies for patients who cannot tolerate CPAP.
Key reflections from the development pathway include the value of staged clinical evaluation, early engagement with healthcare systems, and collaboration between academia, industry, and the NHS. The translational support provided through the HRC framework has enabled the technology to progress from proof-of-concept research to a nationally funded multicentre clinical trial while simultaneously building market awareness and adoption.
The next phase will focus on generating robust clinical evidence and preparing for wider regulatory and healthcare integration. Planned activities include recruitment for the TESLA MICRON UK trial, progression toward Class IIa medical device certification, initiation of regulatory work with the United States Food and Drug Administration, and expansion of international distribution partnerships. Together, these steps aim to support large-scale clinical adoption and establish ZeusOSA as a scalable, non-invasive treatment option for OSA.
Affiliations and links
ZeusOSA has been developed through collaboration between Zeus Sleep Ltd, academic researchers, NHS clinical centres, and patient organisations. The ongoing project is supported by the NIHR HealthTech Research Centre (HRC) in Cardiovascular and Respiratory Medicine and building on results so far, has now been selected by the National Institute for Health and Care Research to run one of the UK’s largest multi centre clinical trials in obstructive sleep apnoea.
Clinical studies are being carried out in NHS sleep centres, with support from the South London Regional Research Delivery Network. Trial design and analysis are supported by King’s Clinical Trials Unit. Patient involvement has been led by Hope2Sleep.
Additional industry partners have supported product development, monitoring, and wider awareness of the technology.