HRC image
The HRC in Devices, digital and robotics plays a key role in identifying unmet clinical needs and generating evidence to support the regulatory approval and adoption of healthcare technologies. The team provides a national service to support the translation of medical devices, digitally delivered health and robotics systems safely and effectively into routine care in the NHS and follow-on social care. They collaborates with industry, academia and healthcare professionals developing technologies across technology readiness levels (TRLs) to improve patient outcomes and experience and improve healthcare efficiency.
About HRC's theme
Medical devices include HealthTech products intended for diagnosing, preventing, monitoring, treating, or alleviating disease and injury that can range in complexity from walking sticks to dialysis machines. Data driven and digitally delivered care refers to the collection, storage, analysis and sharing of patient information and medical metrics such as electronic patient records, wearable data and sensors and telemedicine. Robotics use physical machines and automated systems to perform specific medical or logistical tasks, often with high precision. Digital technologies and robotics can include the integration of artificial intelligence (AI).
Support provided
- Clinical expert reviews: we can provide innovators with expert insight into the unmet clinical need, design and functionality requirements of early-stage health technologies through our clinical expert review process.
- Grant application support: we can help innovators to identify suitable funding opportunities and support grant writing by providing insight and feedback on applications. Working in collaboration with innovators, we can develop work packages to generate evidence to increase technology readiness.
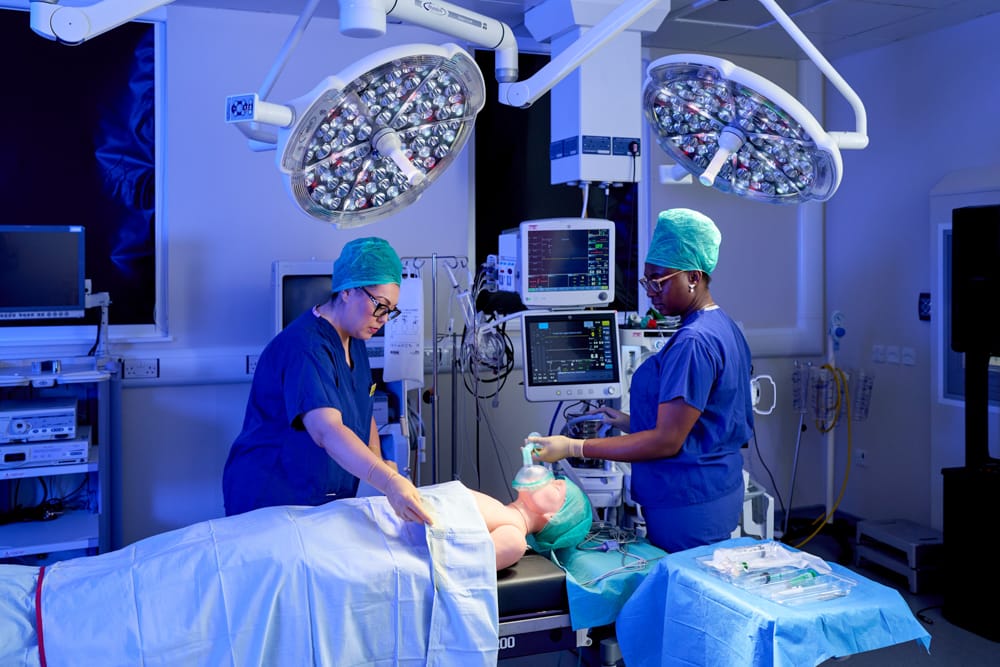
- Human factors and usability testing: we can conduct iterative usability tests across the product lifecycle in realistic environments with patient simulators (working human models) within the MD-TEC facility, in compliance with international standards.
- First-in-human clinical investigations: we can support the preparation of study documentation and submission for regulatory approval to assess essential safety and performance.
- Patient and public involvement and engagement (PPIE): our dedicated PPIE Manager offers enabling support, advice and guidance to help innovators engage effectively with members of the public, ensuring the patient voice is heard throughout technology development.
- Health economics: we develop value proposition calculations, build optimal pricing strategies and conduct model-based economic analyses with support from our dedicated health economist.
- Regulatory support: we understand the appropriate regulations with consideration of evidence generation requirements needed for regulatory approval, where required.
- Data and digital
- Medical devices
- Robotics and autonomous systems
- Human factors and usability testing
- Health economics and value proposition
- Regulation
- Human factors and usability testing
- Health economics
- Patient and public involvement and engagement (PPIE)
- Navigating the regulatory pathway
- Assessment of unmet clinical need
- The NIHR HRC-DDR is co-located with the Medical Devices Testing and Evaluation Centre (MD-TEC). MD-TEC is a dedicated, state-of-the-art medical device and technology usability testing facility that allows the simulation of complex clinical scenarios for product testing
- UHB host a number of NIHR and non-NIHR funded research infrastructures providing opportunities for collaboration. Examples include the NIHR Birmingham Clinical Research Facility, supporting the delivery of clinical investigations and the West Midlands Secure Data Environment (WM-SDE), a resource to support healthcare data needs of technology projects

