HRC Expertise
Each HRC has unique expertise to help you through your journey. However, all HRCs provide support to provide the necessary evidence to support your regulatory approvals.
Specialists in supporting HealthTech across the lifecycle
HRCs are experts in supporting innovators across the product lifecycle to ensure their products are developed safely and with patients at the centre.
While some innovations are funded by larger organisations developing them, the majority of new healthtech innovations come from small to medium sized enterprises (SMEs) or micro-organisations with fewer than 10 employees. In these instances, HRCs typically support the companies to secure the funding necessary to help them move their product along the lifecycle.
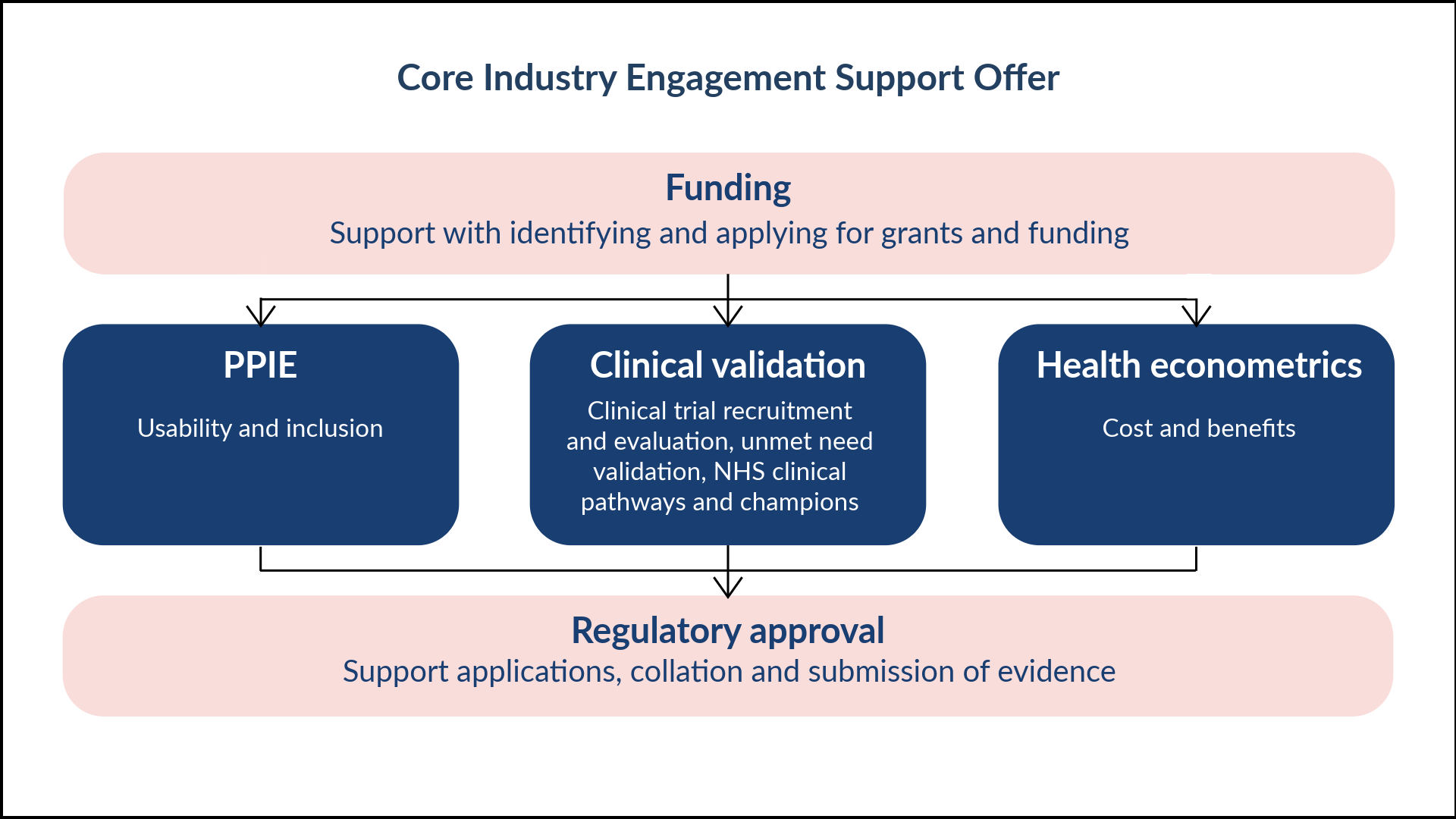
Evidence generation
To apply for regulatory approval to get innovations adopted and used across the NHS, innovators must provide evidence to demonstrate that they have created an innovation that meets a clinical need, that has been developed and trialled with patients, and that demonstrates value to the NHS. Click below to find out more about what types of evidence HRCs can support innovators with.
Many funders now require a full PPIE integration strategy in recognition of the value it provides. From co-design and acceptability testing, through to interpretation of results and advocating for adoption, HRCs can support meaningful PPIE activities to improve the quality of your devices throughout the development process, enhancing your chances of successful adoption and implementation. Find out more here.
Regulatory support
There are many different routes to regulatory approval depending on the type of innovation, and where the innovation will be used. HRCs are experts in guiding innovators to ensure they get the right regulatory approvals to enable them to get their innovation to market and to patients.
- Regulatory classifications
In the UK, healthtech products are classified by the MHRA under the UK MDR 2002 (as amended) based on a tiered, risk-based system that determines the level of regulatory scrutiny required. For software-as-a-medical-device (SaMD) and AI, recent 2026 updates are shifting most diagnostic or treatment-influencing tools out of Class I and into Class IIa or higher, requiring mandatory assessment by a UK Approved Body. For software-as-a-medical-device (SaMD) and AI, recent 2026 updates are shifting most diagnostic or treatment-influencing tools out of Class I and into Class IIa or higher, requiring mandatory assessment by a UK Approved Body.
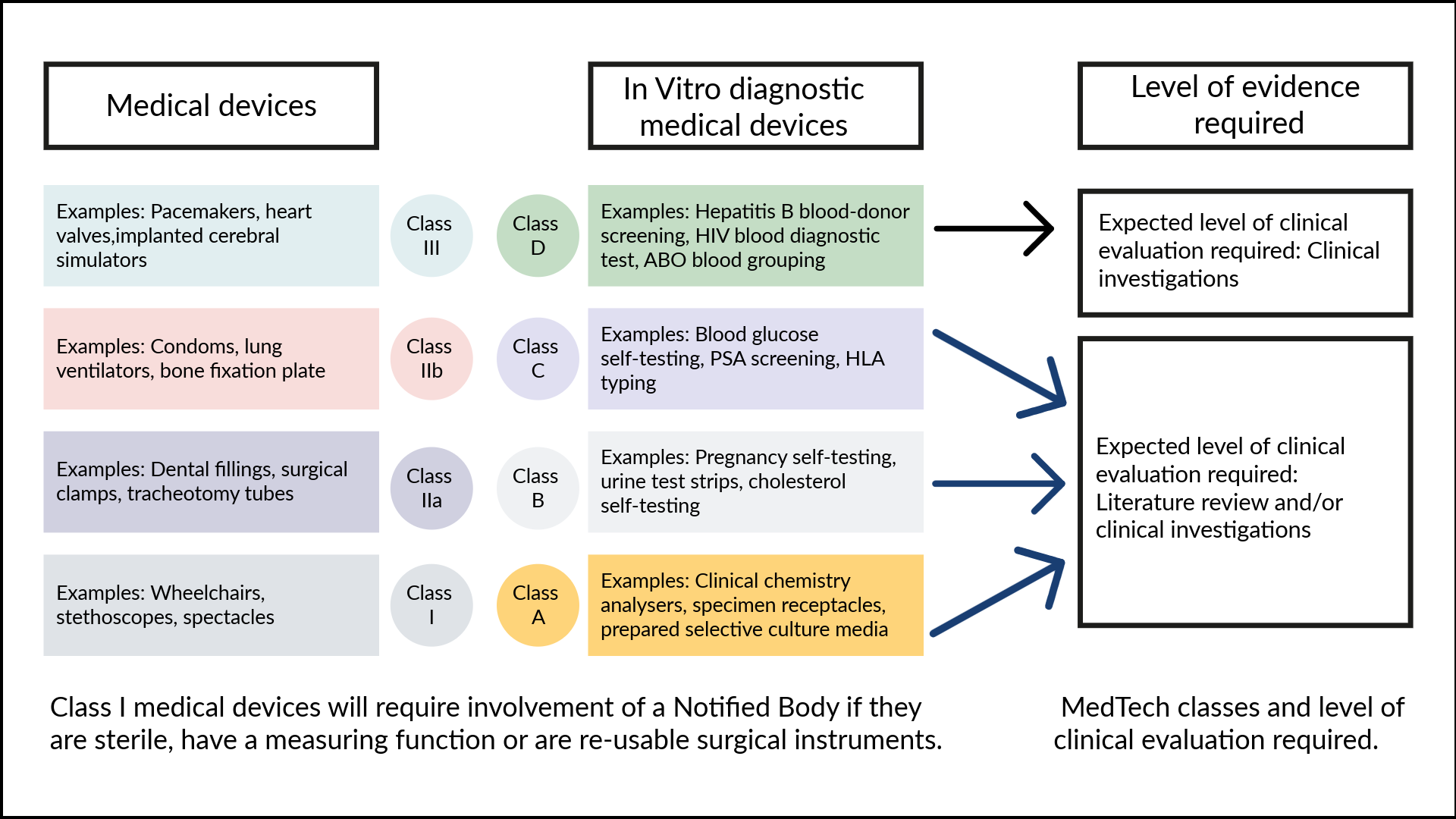
- CE and UKCA regulatory markings
While the UKCA mark remains a valid and available domestic route, the MHRA proposes that devices compliant with the EU MDR and IVDR can be placed on the market without a separate UK certification, a move intended to protect patient access to the 90% of healthtech products currently imported from the EU. Under existing transitional arrangements, CE-marked devices remain legally accepted until at least 2028 or 2030 (depending on the device class), but the new 2026 proposals aim to remove these sunset dates entirely, repositioning the UKCA as a specialized innovation route for first-in-market technologies like AI-as-a-medical-device (AIaMD).
Find the HRC that is right for you
To find out more about each of our HealthTech Research Centres, you can read more about them in the profiles below.
To narrow down your search, you can filter by clinical theme or use the free text box to search for keywords.
Filter by clinical theme
Or type your search term below
Accelerated surgical care
The HRC in Accelerated surgical care are working with medical device, diagnostic and digital health technology companies to develop new innovative medical technologies, generate the evidence to demonstrate financial value (health economics) or improve operational efficiency in the NHS (real-world evidence generation).
They aim to drive HealthTech innovation for patients and healthcare systems to deliver socio-economic benefit through quicker diagnosis and treatment of surgical conditions with safe, early recovery in the community.
Brain health
The HRC in Brain health uses cutting edge technologies including brain scanning, blood sampling, and computerised testing and technology to solve major challenges in the field of brain health and dementia research in diagnosis, care, and treatment. Their aims are to expedite the development and integration of emerging technologies to create a more intelligent and accessible way of diagnosing dementia in the early stages of the condition, and focus on swiftly delivering benefits to patients to reduce the impact of dementia on society.
Brain injury
The HRC in Brain injury supports researchers and innovators to identify unmet needs, evaluate the effectiveness of their technology and implement these solutions to improve the quality of life of those affected by brain injury. Acute care, monitoring of neurological disorders, physical and psychological rehabilitation techniques, and regenerative medicines can all reduce long-term injury and improve long term health. The team works closely with its network of national partners, to support research at every stage of development. They work with patients, carers, and families, to ensure that innovation is meeting a need within the community and making a real difference to those who need it most.
Cardiovascular and respiratory medicine
The HRC in Cardiovascular and respiratory medicine drives innovation and supports the development of HealthTech solutions to address the unmet needs outlined in the NHS Long Term Plan. The team aims to catalyse innovation in HealthTech for the diagnosis and management of cardiovascular and respiratory diseases through cutting-edge technologies and digitally enabled care. This empowers patients and their caregivers to effectively manage cardiovascular health and conditions. They bring together their vast clinical expertise to support researchers and industry throughout the UK to enhance clinical service quality and reduce variation in patient care.

Community healthcare
The HRC in Community healthcare works to improve healthcare outside of hospitals, in the community and interface care, by developing, evaluating and implementing innovative health technologies. Its aim is to improve patient outcomes and reduce pressure on hospital services. The centre operates across community healthcare to identify the clinical needs of clinical staff, patients and carers for technologies that could improve care delivery, life quality and access to care. It works to support the development of appropriate technologies where unmet needs are identified, and robustly evaluates new and existing technologies before deployment, to ensure that they are safe to use and that users understand their strengths and limitations.
Devices, digital and robotics
The HRC in Devices, digital and robotics plays a key role in identifying unmet clinical needs and generating evidence to support the regulatory approval and adoption of healthcare technologies. The team provides a national service to support the translation of medical devices, digitally delivered health and robotics systems safely and effectively into routine care in the NHS and follow-on social care. They collaborates with industry, academia and healthcare professionals developing technologies across technology readiness levels (TRLs) to improve patient outcomes and experience and improve healthcare efficiency.
Diagnostic and technology evaluation
The HRC in Diagnostic and technology evaluation applies innovative clinical research methods to efficiently evaluate diagnostic technologies. The centre examines whether new diagnostics are clinically useful, cost-effective and beneficial to the public. Through supporting funding applications, working in partnership on projects, and delivering training, they help diagnostic innovators thrive and build their evidence. Since 2013 they have worked with over 280 companies including over 180 small and medium-sized enterprises. They have helped innovators obtain over £25 million in external funding and delivered commercially funded projects. They support the development of diagnostic technologies from their invention to their adoption. They specialise in three clinical themes: Ageing and multiple long-term conditions, infection, and precision medicine and rare diseases. By supporting better diagnostics, they hope to improve people’s health and care.

Emergency and acute care
The HRC in Emergency and acute care aims to transform emergency and acute care by developing and deploying new technologies for the detection, diagnosis and treatment of disease, and embedding these technologies into everyday clinical practice. The team work with patients and innovators to co-develop technologies and generate the evidence required to achieve adoption in the health and care system at pace and scale. This ensures new HealthTech solutions are sustainable, benefit all communities, and support fair and equal access to healthcare, helping to improve health and wellbeing for all.

Long-term conditions (Devices for Dignity)
The HRC in Long-term conditions (Devices for Dignity) acts as a catalyst for the development of innovative medical devices, healthcare technologies and technology-dependent interventions that help people to live well for longer. Working alongside patients and carers with lived experience of long-term health conditions, the team accelerates equitable access to HealthTech that has evidenced benefits for people with inter-related conditions, supporting them to live their lives well for longer with impacts on dignity, independence and life quality. Their commitment is to bridge the gap between pioneering research and practical application, ensuring that beneficial healthcare products and transformative interventions reach patients who need them most, improving quality of life and advancing healthcare worldwide

Mental health (MindTech)
The HRC in Mental health (MindTech) is a national centre focusing on the development, adoption and evaluation of new technologies for mental healthcare and dementia. Technology innovation in this area includes diagnosis, monitoring, treatment and support.
The team boasts extensive clinical and research expertise, alongside public involvement groups who bring invaluable lived experience to all their work.

Paediatrics and child health
The HRC in Paediatrics and child health transforms healthcare for children and families by accelerating the development of healthcare technologies. They unite experts in industry, healthcare, and academia in a national child health network, and support the next generation of child health technology innovators through their bespoke education programme. The voices of young people and families are integrated within each research theme through meaningful co-production.
Rehabilitation
The HRC in Rehabilitation brings together researchers, companies, charities, patients and the public to develop technology which helps patients rehabilitate after life changing injury, trauma and illness. The team aims to help patients undergoing rehabilitation recover faster and more fully through the use of technology, supporting NHS staff with better rehabilitation equipment and techniques, and boosting the UK’s rehabilitation technology businesses. The innovation funnel feeds technologies into RehabHRC, taking ideas from technology push and clinical pull, identifying and capturing the most promising technologies for potential RehabHRC support.

Sustainable innovation
The HRC in Sustainable innovation unites researchers, clinicians and medical technology companies to develop, test, and improve medical devices, diagnostics and digital technologies, developing sustainable, innovative, and patient-focused health technologies that will benefit all.
Technological innovations have the potential to revolutionise healthcare services for everyone, helping people to better monitor their health, diagnose ill health sooner and improve management of conditions. In turn this can help to benefit our healthcare systems and the people who work in them. By working collaboratively with businesses we are developing sustainable, innovative, and patient-focused health technologies that improve patient care and benefit the NHS.

